AI-based research takes a step closer to elucidating the cause of Alzheimer's disease, identifying new treatment candidates
Transcriptional regulation by PHGDH drives amyloid pathology in Alzheimer's disease: Cell
https://www.cell.com/cell/fulltext/S0092-8674(25)00397-6
AI Helps Unravel a Cause of Alzheimer's Disease and Identify a Therapeutic Candidate
https://today.ucsd.edu/story/ai-helps-unravel-a-cause-of-alzheimers-disease-and-identify-a-therapeutic-candidate
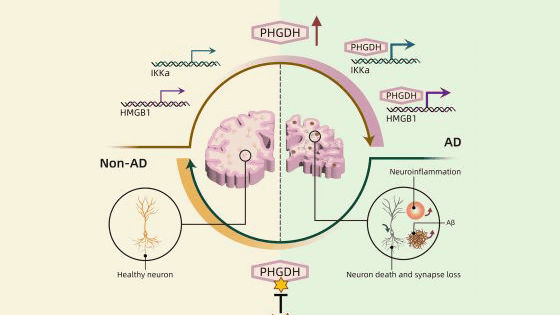
It is believed that Alzheimer's disease may be caused by mutations in certain genes , but these specific genes are only found in a small percentage of all Alzheimer's patients. The research team therefore took a closer look at phosphoglycerate dehydrogenase (PHGDH), a blood biomarker that had been discovered in a 2020 study to be useful for early detection of Alzheimer's disease, and found that the expression level of PHGDH directly correlated with brain changes in Alzheimer's disease. According to the study, the higher the levels of protein and ribonucleic acid (RNA) produced by PHGDH, the more advanced the Alzheimer's disease was.
The research team is further using mouse and human brain organoids to more deeply examine the correlation between PHGDH and Alzheimer's disease. In a paper published in the scientific journal Cell in April 2025, they used AI to visualize the three-dimensional structure of PHGDH and investigate the regulatory role that PHGDH plays in the characteristics of Alzheimer's disease.
As a result, the AI-visualized structure revealed that the substructure of PHGDH was similar to the characteristic shape seen in 'proteins that bind to DNA' ( transcription factors ). This similarity existed only in the structure, not in the protein sequence, so it was important to use AI to construct the 3D structure very accurately, says Sheng Zhong, lead author of the paper.
The research team also demonstrated that PHGDH activates two important target genes, which disrupt the delicate balance of genes and cause a variety of problems that ultimately lead to the early stages of Alzheimer's disease. In short, PHGDH plays a previously unknown role in causing Alzheimer's disease, independent of the enzyme's traditional function.

Furthermore, the structural analysis of PHGDH may also contribute to how Alzheimer's disease can be treated. Many current treatments focus on treating abnormal levels of a sticky protein called beta-amyloid in the brain, but some studies suggest that by that stage it may be ineffective as treatment is too late. However, this discovery could detect the developmental process of Alzheimer's disease at an early stage, so that it can be addressed at the earliest stage when beta-amyloid starts to accumulate.
The team again used AI to perform 3D visualization and modeling to confirm how NCT-503, which is believed to be effective in inhibiting the enzymatic activity of PHGDH, accesses the DNA-binding substructure of PHGDH. In the study, they tested NCT-503 in two mouse models of Alzheimer's disease and showed that it significantly alleviated the progression of the disease.
'We now have a therapeutic candidate with proven efficacy that has the potential to progress to clinical trials for use in humans,' said lead author Sheng Zhong, who spoke about the significance of the discovery and future research. 'Furthermore, our study suggests that completely different types of small molecules may also be useful in future drug development.'
Related Posts:
in Science, Posted by log1e_dh